Sanitation

RReady to Build a Stronger Food Safety Program?
Sanitation Development & Support
A clean and hygienic production environment is the backbone of food safety and quality. Effective sanitation practices prevent contamination, ensure compliance with regulatory standards, and support operational efficiency.
At Points North Certified, we specialize in designing and implementing tailored sanitation systems that meet the highest industry standards and regulatory requirements.
Our comprehensive approach helps your facility maintain a safe, clean, and compliant environment every day.
Why Is Sanitation Critical?
A comprehensive and well-maintained sanitation system is the cornerstone of food safety and operational excellence. It not only ensures your facility adheres to regulatory requirements but also directly impacts your brand reputation and consumer trust.
Implementing an effective sanitation program helps prevent cross-contamination, physical debris, and microbial growth, which are common sources of foodborne illnesses and safety issues.
What is Sanitation?
Sanitation involves the systematic cleaning and disinfecting of your facility, equipment, and work surfaces to prevent product contamination and ensure food safety. A well-structured sanitation program is essential to meet industry regulations and safeguard consumer health.
Structured Procedures & Validation
Our sanitation approach includes developing detailed procedures, validation protocols, and documentation to verify the effectiveness of cleaning processes.
Regular inspection and validation ensure hygiene practices are maintained consistently and effectively, reducing risks and supporting audit readiness.
Support & Continuous Improvement
We deliver a fully customizable sanitation framework equipped with operational tools, checklists, and schedules to ensure daily execution, inspections, and ongoing improvement.
This comprehensive system helps your sanitation team perform verifiable hygiene practices, maintain product integrity, and meet audit and compliance requirements.
Wherever You Are in the Process, We've Got You Covered

Creation
We can create your Food Defense aligned with industry standards, including GFSI-recognized schemes, USDA, and FDA regulatory requirements.
Our tailored solutions provide a strong foundation for compliance, operational efficiency, and industry performance, ensuring your programs are scalable and audit-ready.

Gap Analysis
Our thorough assessments identify weaknesses in your current processes and systems, whether related to GFSI standards, USDA, or FDA regulations.
By pinpointing vulnerabilities early, we enable you to proactively address gaps, strengthen your compliance posture, and prepare effectively for audits or certification processes.

Validation
We confirm that your controls, processes, and systems are effective through science-based validation tailored to GFSI schemes, USDA, or FDA standards.
Our validation services help reduce risks, demonstrate ongoing compliance, and support continuous improvement in your food safety and quality management systems.
Implementation
Our team supports seamless integration of new or existing programs, ensuring they meet industry standards such as GFSI, USDA, and FDA.
We assist in training staff, establishing procedures, and embedding controls into daily operations—helping you achieve system integrity and regulatory readiness.
Training & Support
We empower your team with customized training programs focused on GFSI, USDA, or FDA compliance requirements.
Our ongoing support ensures your programs are maintained properly, fostering a strong culture of food safety, continuous improvement, and regulatory adherence.
Creation
We can create your Food Defense aligned with industry standards, including GFSI-recognized schemes, USDA, and FDA regulatory requirements.
Our tailored solutions provide a strong foundation for compliance, operational efficiency, and industry performance, ensuring your programs are scalable and audit-ready.
Gap Analysis
Our thorough assessments identify weaknesses in your current processes and systems, whether related to GFSI standards, USDA, or FDA regulations.
By pinpointing vulnerabilities early, we enable you to proactively address gaps, strengthen your compliance posture, and prepare effectively for audits or certification processes.
Sanitation Documentation Suite
At Points North Certified, we provide a comprehensive set of sanitation documents designed to establish, maintain, and verify your facility’s hygiene and cleanliness standards. These documents support compliance with industry regulations, enhance operational consistency, and foster a culture of food safety excellence.
Our Sanitation Documents
Sanitation Programs
A detailed plan outlining your facility’s overall sanitation objectives, procedures, and responsibilities tailored to meet regulatory standards and best practices.
Sanitation Schedules
A structured timetable that defines when and how cleaning tasks should be performed across all areas and equipment, ensuring routine compliance and hygiene.
Sanitation Checklists
Step-by-step lists are used to verify that sanitation tasks are completed correctly, providing a record of routine cleaning activities for accountability and traceability.
SSOPs for All Equipment
Sanitation Standard Operating Procedures are specifically created for each piece of equipment, ensuring effective cleaning and sanitization to prevent cross-contamination.
Task Checklists
Detailed checklists for individual sanitation tasks, assigned to specific team members to ensure all steps are followed precisely and nothing is overlooked.
Routine Inspection Reports
Includes pre-Op and post-Op sanitation checklists used during routine inspections to verify ongoing hygiene standards before and after production processes.
Zone Identification Report
A report designating different areas or zones within your facility based on risk level, helping focus sanitation efforts where they are most needed, and ensuring targeted cleaning protocols.
Helpful Links
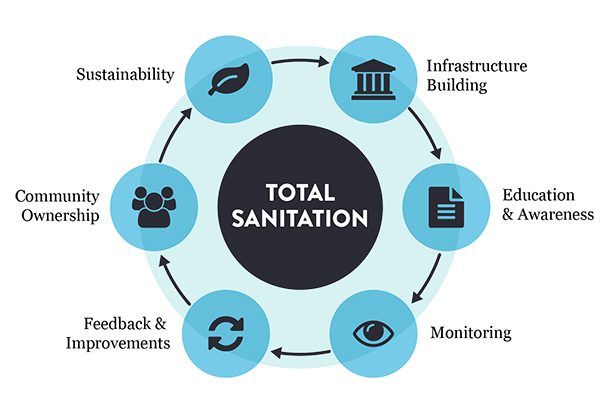
Why Partner with Points North Certified?
Our relationship doesn’t end once your programs are created. We offer ongoing support, help monitor performance, and review your systems periodically to identify areas for improvement, ensuring your food safety practices stay current and effective.
Experienced Industry Experts
Our team brings decades of hands-on validation experience across multiple food sectors, including meat & poultry, baked goods, snack foods, beverages, and more.
Regulatory & Certification-Ready
We tailor validation procedures to meet FDA, USDA, GFSI, and other relevant standards, ensuring your processes are audit-ready and certification compliant.
Detailed and Clear Documentation
Our validation reports and protocols are comprehensive, transparent, and designed to support your internal records, external audits, and regulatory inspections.
Support & Continual Improvement
We provide guidance for ongoing validation activities, re-validation schedules, and performance monitoring to sustain food safety over time.

